
 Chenghui Shuangda
Chenghui Shuangda

Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. proudly announces that its self-developed Active Pharmaceutical Ingredient (API), Apalutamide (Registration No.: Y20240001176), has successfully passed the review by the National Medical Products Administration (NMPA) on March 12, 2026. The company has officially obtained the Category A certification (approval for APIs used in marketed formulations). This marks a critical leap from R&D to commercial application for the product!
The successful approval of Apalutamide API is a significant achievement in the company's strategic layout for innovative drug R&D and industrialization. It fully demonstrates our profound technical expertise and robust industrialization capabilities in the field of innovative drug development. From the laboratory research stage to large-scale production, we have strictly adhered to international GMP standards to ensure comprehensive quality control across the entire process.
We now sincerely invite global pharmaceutical enterprises, research institutions, and supply chain partners to explore strategic cooperation. Let us join hands to capitalize on the market opportunities presented by this innovative achievement and work together to drive the high-quality development of the pharmaceutical industry.
Product Profile
Product Name: Apalutamide
English Name
CAS No.: 956104-40-8
Molecular Formula: C21H15F4N5O2S
Structural Formula:
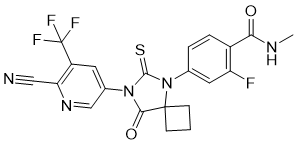
Application Overview:
Apalutamide is indicated for the treatment of Non-Metastatic Castration-Resistant Prostate Cancer (NM-CRPC). As a second-generation, highly selective androgen receptor inhibitor, it significantly prolongs the metastasis-free survival of patients.